Covid Vaccines and PIMS-TS
Within our PIMS-TS family support group of 3.3K+ members, there is a differing range of opinions on the efficacy of Covid vaccines in children generally, and in relation to PIMS-TS children specifically. This is as it should be and reflects the views of society in general. We hope that the following discussion is measured and looks at this question from all viewpoints. Please note that the graphs and statistics are from a variety of sources and that we cannot verify their accuracy.
For those parents wanting to be given the choice of COVID vaccination for their children, the long, hesitant wait in the UK has been frustrating. If indeed vaccines could have prevented at least some of the PIMS-TS reactions in children, then they are arriving too late, as an estimated 80% of children have now been infected by COVID-19, with 50% of these through the Omicron variant. These statistics are from the JCVI statement on elective vaccination of 5-11 year-olds in the UK - see link below to full document. The document also puts forward the statistical benefits of the vaccine in potential mild or medium COVID outbreaks in the future.
JCVI statement: Statement-on-vaccination-of-children-aged-5-to-11-years-old
Dr. Deepti Gurdasani, clinical epidemiologist said:
"The JCVI report on vaccination of 5-11 year-olds says that they estimate that about half of 5-11 year-olds likely got infected during the omicron wave. Yet, while they could've offered vaccines to children then, they delayed. And delayed. And let them get mass infected."
So, these are some of the frequently asked questions surrounding vaccines and PIMS-TS:
1. Could the vaccine reduce the risk of, or prevent PIMS-TS?
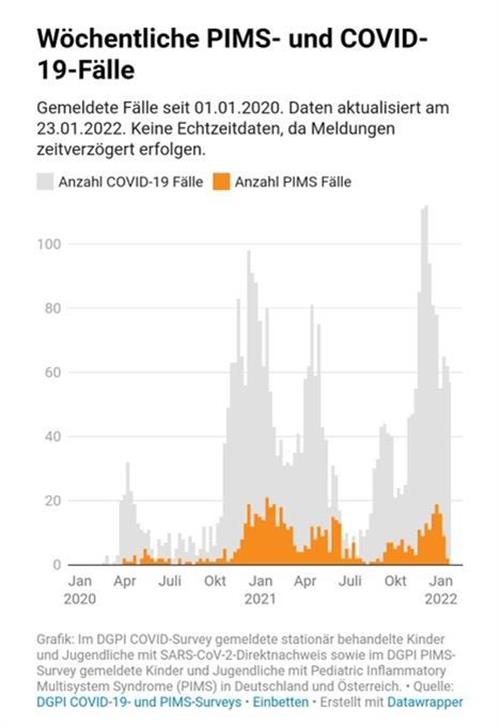
The rationale is that by giving children a controlled dose of a non-live vaccine, the child will produce antibodies to COVID-19 in a calmer way than it might if confronted with a live COVID-19 infection. The vaccine itself should not, in theory, provoke an auto inflammatory reaction. It should then prevent an auto inflammatory reaction should the child subsequently be infected with COVID-19, as the body should have developed antibodies that will recognise COVID infection and respond to it in a measured way. Many countries have been offering children and young people COVID vaccines for longer than we have in the UK. Research has come out from countries such as the USA and France, showing that the number of PIMS-TS / MIS-C cases have fallen since the vaccines have been introduced. The graph below from the USA seems to confirm this; however, it is not up to date and doesn’t fully represent the Omicron wave. We could also ask: was the Omicron variant perhaps not such a trigger for PIMS-TS than Alpha or Delta? Alternatively, if the majority of children and young people had already been exposed to COVID-19 by the end of 2021, the percentage of children that will continue to develop PIMS-TS is likely to be much smaller. It is very unusual to get PIMS-TS twice, so cases will reduce over time, as long as variants do not change this pattern. To explore these questions, we need to look carefully into the studies carried out on this matter and at the data. Parents of PIMS-TS children might decide that potentially genetically similar siblings, particularly those who have not yet had COVID-19, might be at a higher risk of PIMS-TS and choose to vaccinate. The decision about vaccination of PIMS-TS children to prevent PIMS-TS is perhaps less urgent, as children are unlikely to get PIMS-TS again. Parents still then need to consider other reasons for vaccination, for example the potential for developing Long COVID symptoms or indeed the possibility of becoming ill from COVID-19 itself.
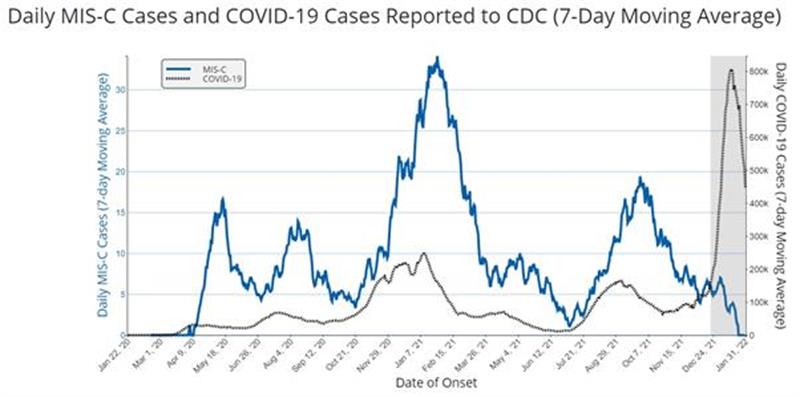
CDC MIS-C site with graph showing a dramatic drop in PIMS-TS cases after the vaccine rollout in children.

Research from the USA on vaccines and reduction in MIS-C: CDC.gov-volumes-71wr/mm7102e1.htm
Research from France on vaccines and reduction in MIS-C: Jama-levy-2021-90855.pdf
2. Could the vaccine trigger PIMS-TS?
When vaccines were first being trialled, PIMS-TS was included in the draft copy small print as a possible, if unlikely, adverse event. It was one of the things they needed to investigate, and quite rightly. Because PIMS-TS is an inflammatory response, even a non-live vaccine could potentially trigger it. And if the plan was to roll out the offer of vaccines to all children globally, it was important to ensure the trials were thorough. Details of the initial vaccine trials are not easy to come by, but we hope that the PIMS-TS connection was covered, by trialling with a large enough group of child volunteers to allow for the small percentage who might have a genetic predisposition to PIMS-TS.
As far as we are aware, none of the vaccines approved for children and young people include PIMS-TS as a contraindication on their parent information leaflet.
There are a few cases of PIMS-V / MIS-V reported, and many countries have been watching out for this after vaccine rollout. The numbers appear to be very small, a search on the internet shows two cases in children and one in an adult, which is reassuring.
Pfizer do not list PIMS-TS / MIS-C as one of possible adverse events to the vaccine or mention any cases occurring in their trials. But it is listed as a post vaccine rollout possible adverse event that must be mandatory reported through the VAERS system in the USA and voluntary reporting is suggested through the yellow card system in the UK. Labeling.pfizer.com/ShowLabeling=pdf
The UK yellow card reporting system vaccine analysis print for Pfizer, reports 6 cases of Systematic inflammatory response syndrome: COVID-19_Pfizer-BioNTech_Vaccine_Analysis_Print.pdf
Report of a case of MIS-V in an adult: BMJ.com/content/14/7/e243888
Preprint research from France on vaccine induced MIS-C: Medrxiv.org-content-10.1101/2022
An article from Spain, looking both at the risk of vaccine induced SIMP (sindrome-inflamatorio-multisistemico-pediatrico - ie. PIMS) and the suitability of COVID vaccines for children who have had SIMP. Interestingly, they put forward the idea that SIMP might be an autoimmune response rather than an autoinflammatory one: Vacunasaep.org-profesionales-noticias-vacunas-covid19-y-SIMP
Vaccination of children with a history of MIS-C
3. Can a child get PIMS-TS more than once?
It is extremely unlikely, just as it is with Kawasaki Disease, but not unheard of. Sometimes, an apparent recurrence can happen soon after initial infection, displaying some of the same symptoms, but not so acutely. In some children there seems to be a susceptibility to autoinflammatory responses in general and research is being carried out on this. Some consultants are running blood tests for children who have had PIMS-TS, four weeks after a subsequent infection with COVID as a precaution and to contribute to research in this area.
4. Are siblings more likely to get PIMS-TS?
Potentially yes, as there is a genetic predisposition involved in PIMS-TS, as in Kawasaki Disease. Siblings who have not had COVID-19 or PIMS-TS might consider vaccination for this reason. Also, parents of PIMS-TS children, as PIMS-TS can affect adults in rare cases and is referred to as MIS-A or PIMS-TS in adults.
5. Are there any groups of children who are more susceptible to PIMS?
Yes, more boys than girls (3 to 2), and more in children of Black, Hispanic/Latino and Asian or Pacific Islander ethnicities.
6. Are there other reasons children should have the COVID vaccine?
PIMS-TS is not the only condition caused by COVID-19. Long COVID is common in children and is chronic and long lasting compared to the acute but generally shorter lasting PIMS-TS. The COVID vaccine lessons the likelihood of contracting COVID, but breakthrough cases might still result in Long COVID. Omicron seems to infect children multiple times, perhaps increasing or compounding the chance of Long COVID symptoms. Notably, some children with Long COVID who have the vaccine afterwards find that it reduces their Long COVID symptoms either in the short or longer term. There is ongoing research in this area.
Plus, COVID infection itself is not without risk in children, especially in CV and CEV children or in children with other underlying health problems. Children with family members who are CEV might choose to be vaccinated to protect their family.
Further Reading:
7. Will the vaccine protect children against PIMS-TS triggered by any new variants?
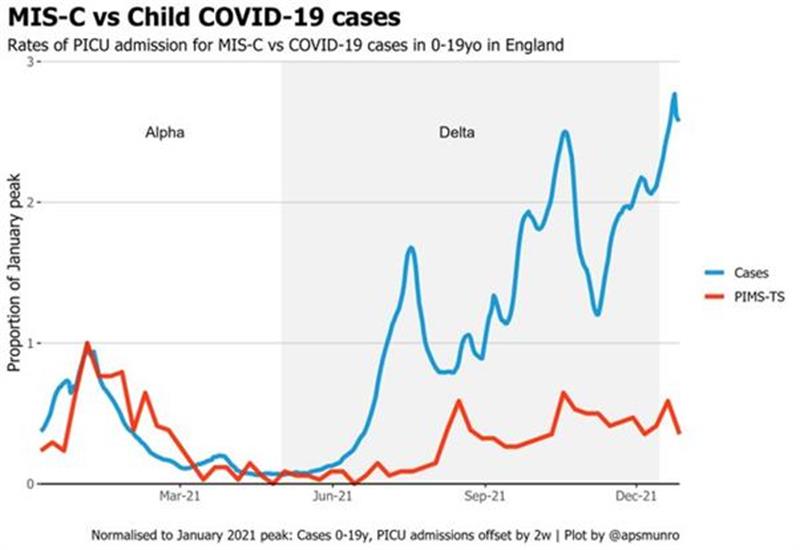
This is a difficult question to answer. PIMS-T / MIS-C cases seem to have been less with Omicron than Alpha or Delta in the USA. The vaccination roll out in children and young people might explain this, as might the high number of children who have now had COVID-19. The UK has however, unofficially (there is no public PIMS data) said that the numbers of PIMS-TS cases have been less with Delta than Alpha and even less with Omicron. In the UK, the vaccine rollout in children and young people has been slower than the USA and other countries. Does this point to vaccines or variants, or most children having already had COVID as the reason for the drop in PIMS cases, assuming they have dropped? Is anyone looking at the data in the UK to verify this? We are in a unique position to do this, given our late roll out of child vaccines compared with other countries. It would in our view be insightful research.
Could it also be that because of the UK’s lack of mitigations to prevent infection compared with other countries, that more children got PIMS-TS early on and that is why the cases are down with Delta and even more down on Omicron? It is estimated that in the UK, 85% of all 5-11 year-olds have experienced COVID infection, 50% of which from the Omicron variant.
Cases of breakthrough infections in fully vaccinated adolescent patients.
General Vaccination advice for children who have had PIMS-TS.
There is guidance as to how long to wait before any vaccinations after PIMS-TS and whether live vaccines such as the flu nasal spray need to be avoided. For more information on this please follow this link to the section in Follow up Care.
Vaccination Post Kawasaki Disease
The British Medical Journal article (BMJ-content/86/4/286.long) says:
"There are two important issues relating to vaccination following an episode of KD. Firstly, IVIG can block replication of live viral vaccines and subsequent actively acquired immunity. Consequently it is currently recommended that live vaccines be deferred for at least three months following treatment with IVIG. Secondly, an important question remaining unanswered is the safety of live or other vaccines in children recovering from KD. It has been our observation that some autoimmune disease states, including the systemic vasculitides, flare in response to live and non-live vaccine preparations. It is therefore our policy to defer immunisation with all vaccines for at least three months following an episode of KD."